Introduction to radioactivity and radioactive material
Introduction to Radioactivity & Radioactive material
Radioactivity is the Spontaneous emission of radiation, either directly from unstable atomic nuclei or as a consequence of a nuclear reaction. The radiation, including alpha particles, nucleons, electrons, and gamma rays, emitted by a radioactive substance.
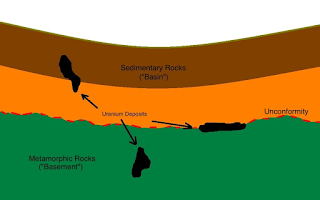
A Closer Look: In the nuclei of stable atoms, such as those of lead, the force binding the protons and neutrons to each other individually is great enough to hold together each nucleus as a whole. In other atoms, especially heavy ones such as those of uranium, this energy is insufficient, and the nuclei are unstable. An unstable nucleus spontaneously emits particles and energy in a process known as radioactive decay. The term radioactivity refers to the particles emitted. When enough particles and energy have been emitted to create a new, stable nucleus (often the nucleus of an entirely different element), radioactivity ceases. Uranium 238, is a very unstable element, goes through 18 stages of decay before becoming a stable isotope of lead, lead 206. Some of the intermediate stages include the heavier elements thorium, radium, radon, and polonium.
All known elements with atomic-numbers greater than 83 (bismuth) are radioactive, and many isotopes of elements with lower atomic numbers are also radioactive. When the nuclei of isotopes that are not naturally radioactive are bombarded with high-energy particles, the result is artificial radioisotopes that decay in the same manner as natural isotopes. Each element remains radioactive for a characteristic length of time, ranging from mere microseconds to billions of years. An element's rate of decay called half-life. This refers to the average length of time it takes for half of its nuclei to decay.
Alpha, Beta, & Gamma Rays
The extra energy, or radiation, emitted by radioactive elements comes in three different types: alpha, beta, and gamma.
Alpha radiation is a stream of alpha particles, which are positively charged. They're fairly large, which means they have a difficult time getting through materials like clothes and paper.
Beta radiation is a stream of beta particles, which are negatively charged. These particles can more easily penetrate materials like clothes, and they can even get deep into your skin where they can do harm to your cells. But they can be blocked with denser materials like aluminium.
Gamma radiation is high-frequency electromagnetic radiation. Gamma rays have no charge but have A LOT of energy; more energy than even visible light or X-rays. Because of this, they are able to pass right through most materials, making them quite dangerous. But they can't penetrate very dense materials like lead, which is why you may be given a lead vest for protection in a hospital or laboratory.
Radioactive Materials Are Natural
Most people think of radioactive materials as harmful, man-made materials, but quite the opposite is true. In fact, most radioactive materials occur naturally in the environment and have been around much longer than humans!
Most of the radiation we're exposed to on Earth comes from the sun and stars in outer space. We are constantly bombarded with this radiation but we are partially protected from it by the atmosphere around us. Those at sea level are more protected than those at higher altitudes because the protective atmosphere thins with increasing elevation.
Radiation also comes from other things like rocks and minerals. Because of this, those who live in houses made of brick, concrete, or stone receive a greater amount of radiation than those who live in houses made of wood.
Geiger muller counter
The Geiger counter is an instrument used for measuring ionizing radiation
It detects ionizing radiation such as alpha particles, beta particles and gamma rays using the ionization effect produced in a Geiger–Müller tube
It is perhaps one of the world's best-known radiation detection instruments
It detects ionizing radiation such as alpha particles, beta particles and gamma rays using the ionization effect produced in a Geiger–Müller tube
It is perhaps one of the world's best-known radiation detection instruments










Comments
Post a Comment